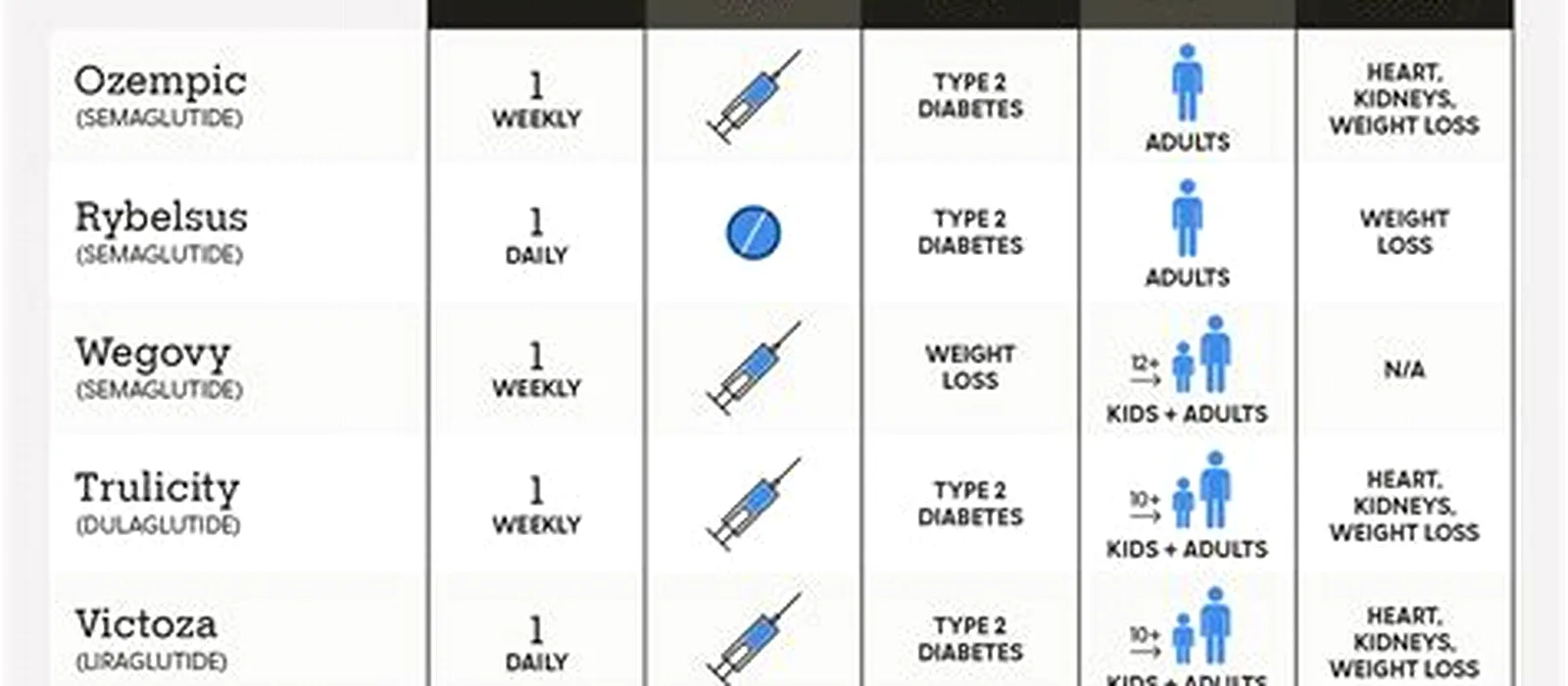
Overview of FDA-approved GLP-1 medications
The GLP-1 drug class has expanded substantially since the first approval in 2005. Today's options differ in:
- FDA indication: some are approved for type 2 diabetes, others for weight management, and a few for both (under different brand names).
- Route: primarily subcutaneous injection, with one oral tablet (Rybelsus).
- Dosing frequency: twice-daily, once-daily, or once-weekly.
- Receptor selectivity: single GLP-1R agonist versus dual GIP/GLP-1R agonist (tirzepatide).
- Evidence base: mean weight loss ranges from about 5% (older agents at older doses) to 21% (tirzepatide 15 mg).
Below is each FDA-approved drug, with brand names, manufacturer, indications, dosing, and the key clinical evidence.
| Brand | Generic | Manufacturer | Frequency | FDA indication |
|---|---|---|---|---|
| Ozempic | Semaglutide | Novo Nordisk | Once weekly (Ozempic, Wegovy); once daily (Rybelsus oral) | Type 2 diabetes (FDA-approved 2017); cardiovascular risk reduction in adults with T2D |
| Wegovy | Semaglutide | Novo Nordisk | Once weekly (Ozempic, Wegovy); once daily (Rybelsus oral) | Chronic weight management (BMI ≥30, or ≥27 with comorbidity); FDA-approved 2021; CV risk reduction with overweight/obesity and known heart disease |
| Rybelsus | Semaglutide | Novo Nordisk | Once weekly (Ozempic, Wegovy); once daily (Rybelsus oral) | Type 2 diabetes; oral form, FDA-approved 2019 |
| Mounjaro | Tirzepatide | Eli Lilly | Once weekly | Type 2 diabetes; FDA-approved 2022 |
| Zepbound | Tirzepatide | Eli Lilly | Once weekly | Chronic weight management; FDA-approved November 2023; obstructive sleep apnea in obesity (approved December 2024) |
| Saxenda | Liraglutide | Novo Nordisk | Once daily | Chronic weight management; FDA-approved 2014 |
| Victoza | Liraglutide | Novo Nordisk | Once daily | Type 2 diabetes; FDA-approved 2010 |
| Trulicity | Dulaglutide | Eli Lilly | Once weekly | Type 2 diabetes; FDA-approved 2014 |
| Byetta | Exenatide | AstraZeneca | Twice daily (Byetta) or once weekly (Bydureon) | Type 2 diabetes; FDA-approved 2005 |
| Bydureon BCise | Exenatide | AstraZeneca | Twice daily (Byetta) or once weekly (Bydureon) | Type 2 diabetes; extended-release, FDA-approved 2012 |
Semaglutide: Ozempic, Wegovy, Rybelsus
Semaglutide is the GLP-1 drug with the largest published evidence base and the most name recognition. Manufactured by Novo Nordisk.
Ozempic (semaglutide injection, type 2 diabetes)
FDA-approved 2017 for adults with type 2 diabetes as adjunct to diet and exercise to improve glycemic control. Approved in 2020 for reduction of major adverse cardiovascular events in adults with T2D and established cardiovascular disease, based on the SUSTAIN-6 trial. Once-weekly subcutaneous injection at doses of 0.25, 0.5, 1.0, or 2.0 mg.
Wegovy (semaglutide injection, weight management)
FDA-approved June 2021 for chronic weight management in adults with BMI ≥30, or ≥27 with at least one weight-related comorbidity. Approved December 2022 in adolescents 12 and older with BMI at or above the 95th percentile for age and sex. Approved March 2024 for cardiovascular risk reduction in adults with established cardiovascular disease and overweight or obesity, based on the SELECT trial (16,000+ participants, 20% reduction in MACE). Once-weekly injection, titrated over 16-20 weeks to a maintenance dose of 2.4 mg.
Rybelsus (oral semaglutide, type 2 diabetes)
FDA-approved September 2019 for type 2 diabetes. The first and currently only FDA-approved oral GLP-1 receptor agonist tablet. Once-daily, taken on an empty stomach with a small amount of water. Doses of 3, 7, or 14 mg. Oral bioavailability is achieved by formulating semaglutide with the absorption enhancer SNAC (salcaprozate sodium). Weight loss with Rybelsus is meaningful but smaller than injectable semaglutide.
Semaglutide evidence base
The STEP trials (Semaglutide Treatment Effect in People with obesity), published 2021-2022 in NEJM and Lancet, established semaglutide 2.4 mg weekly as a chronic weight management drug. STEP-1 reported 14.9% mean weight loss at 68 weeks in adults without diabetes; STEP-2 in adults with T2D reported ~9.6%. The SELECT trial (2023) extended the evidence into cardiovascular outcomes.
Tirzepatide: Mounjaro and Zepbound
Tirzepatide is the first FDA-approved dual GIP/GLP-1 receptor agonist. Manufactured by Eli Lilly.
Mounjaro (tirzepatide injection, type 2 diabetes)
FDA-approved May 2022 for type 2 diabetes. Once-weekly subcutaneous injection. Doses of 2.5, 5, 7.5, 10, 12.5, or 15 mg, titrated by 2.5 mg every four weeks based on tolerance.
Zepbound (tirzepatide injection, weight management)
FDA-approved November 2023 for chronic weight management with the same BMI criteria as Wegovy. FDA-approved December 2024 for moderate-to-severe obstructive sleep apnea in adults with obesity, based on the SURMOUNT-OSA trial. Same dose range as Mounjaro.
Tirzepatide evidence base
The SURMOUNT trial series demonstrated the largest published weight loss in any obesity drug trial. SURMOUNT-1 (2022): 20.9% mean weight loss at 72 weeks on 15 mg in adults without diabetes. SURMOUNT-2 in T2D: ~14.7%. In a head-to-head trial against semaglutide (SURMOUNT-5, 2025), tirzepatide produced greater weight loss than semaglutide 2.4 mg.
Liraglutide: Saxenda and Victoza
Manufactured by Novo Nordisk. Once-daily subcutaneous injection — older dosing schedule than the once-weekly agents.
Victoza (liraglutide, type 2 diabetes)
FDA-approved 2010 for type 2 diabetes. Doses of 0.6 mg, 1.2 mg, or 1.8 mg daily.
Saxenda (liraglutide, weight management)
FDA-approved 2014 for chronic weight management at a higher dose (3.0 mg daily) than Victoza. The SCALE-Obesity trial reported mean weight loss of 8.0% at 56 weeks — meaningful but smaller than semaglutide or tirzepatide.
Liraglutide is sometimes preferred in specific clinical situations (e.g., when daily dose flexibility is helpful) but is less commonly chosen for new patients now that more effective once-weekly options exist.
Dulaglutide: Trulicity
Manufactured by Eli Lilly. Once-weekly subcutaneous injection in a single-use pen device. FDA-approved 2014 for type 2 diabetes; in 2020 for reduction of cardiovascular events in adults with T2D, based on the REWIND trial. Doses of 0.75, 1.5, 3.0, or 4.5 mg weekly. No FDA-approved weight management indication.
Exenatide: Byetta and Bydureon
Manufactured by AstraZeneca (originally Amylin/Eli Lilly). Byetta (5 or 10 mcg twice-daily) was the first GLP-1 receptor agonist approved (2005). Bydureon BCise (2 mg weekly extended-release) followed in 2012. Both are approved for type 2 diabetes; not approved for weight management. Less commonly prescribed today.
Compare options with a licensed provider
A clinician can review your medical history, insurance, and goals to recommend the GLP-1 medication that fits your situation.
See If You Qualify →Sponsored link. We earn a commission when you sign up. Medical eligibility determined by licensed providers. Disclosure
Compounded GLP-1 medications
Beyond FDA-approved brand-name products, several compounding pharmacies prepare semaglutide and tirzepatide on a per-patient basis under section 503A of the Federal Food, Drug, and Cosmetic Act. The FDA's role with compounded drugs is regulating the pharmacies (state and federal), not approving the compounded products themselves.
Legal availability of compounded semaglutide and tirzepatide changes with FDA drug shortage status. Both products have been on and off the FDA shortage list at various points in 2024-2026. After a drug is removed from the shortage list, 503A compounding is permitted only when there is a specific clinical need that the commercial product cannot meet — for example, a dose not commercially available, or an excipient allergy. State boards of pharmacy may add additional restrictions.
For details on the current status and how telehealth platforms work with compounding pharmacies, see compounded semaglutide and compounded tirzepatide.
Emerging GLP-1 agents (not FDA-approved as of review date)
- Retatrutide (Eli Lilly): triple GLP-1/GIP/glucagon receptor agonist; Phase 3 obesity and T2D trials. Phase 2 data showed ~24% mean weight loss at 48 weeks.
- Orforglipron (Eli Lilly): oral small-molecule GLP-1 receptor agonist; Phase 3 ATTAIN and ACHIEVE trials. Important because it is a non-peptide oral, potentially manufacturable at lower cost.
- CagriSema (Novo Nordisk): combination of semaglutide and cagrilintide (an amylin analog). Phase 3 trials in obesity.
- Survodutide (Boehringer Ingelheim / Zealand): dual GLP-1/glucagon agonist; Phase 3 obesity trials.
How clinicians choose between GLP-1 medications
Decisions typically consider:
- FDA indication match (T2D vs weight management vs CV risk reduction).
- Magnitude of effect needed (tirzepatide produces the largest mean weight loss; semaglutide is well-studied for cardiovascular benefit).
- Insurance formulary coverage.
- Dosing preference (weekly injection vs daily oral).
- Tolerability history with prior GLP-1 trials.
- Contraindications (thyroid history, pancreatitis history, pregnancy planning).
- Cost in the patient's specific scenario.
The "best" GLP-1 medication is not a single answer — it depends on the individual case. A licensed clinician is the appropriate person to weigh these factors with you.
Find your GLP-1 option
A licensed clinician via telehealth can evaluate your eligibility for brand-name or legally compounded GLP-1 medications.
Check Your Eligibility →Sponsored link. We earn a commission when you sign up. Medical eligibility determined by licensed providers. Disclosure