What is GLP-1?
GLP-1 stands for glucagon-like peptide-1, an incretin hormone secreted by L-cells in your small intestine after you eat. It acts on multiple organs: the pancreas (stimulating glucose-dependent insulin secretion and suppressing glucagon), the stomach (slowing gastric emptying), and the brain (signaling satiety in the hypothalamus and brainstem).
Natural GLP-1 has a half-life of about two minutes — it is rapidly degraded by the enzyme dipeptidyl peptidase-4 (DPP-4). The breakthrough behind GLP-1 medications was engineering synthetic analogs that bind the same GLP-1 receptor (GLP-1R) but resist DPP-4 degradation, extending their half-life from minutes to hours (for daily drugs like liraglutide) or days (for weekly drugs like semaglutide and tirzepatide).
The first GLP-1 receptor agonist, exenatide (Byetta), was derived from exendin-4, a peptide found in the saliva of the Gila monster (Heloderma suspectum). It was approved by the FDA in 2005 for type 2 diabetes. Every GLP-1 drug since has built on that template, with semaglutide and tirzepatide producing the largest weight loss effects in clinical trials.
FDA-Approved GLP-1 Medications
As of 2026, the following GLP-1 receptor agonist active ingredients are FDA-approved in the United States. All are prescription-only.
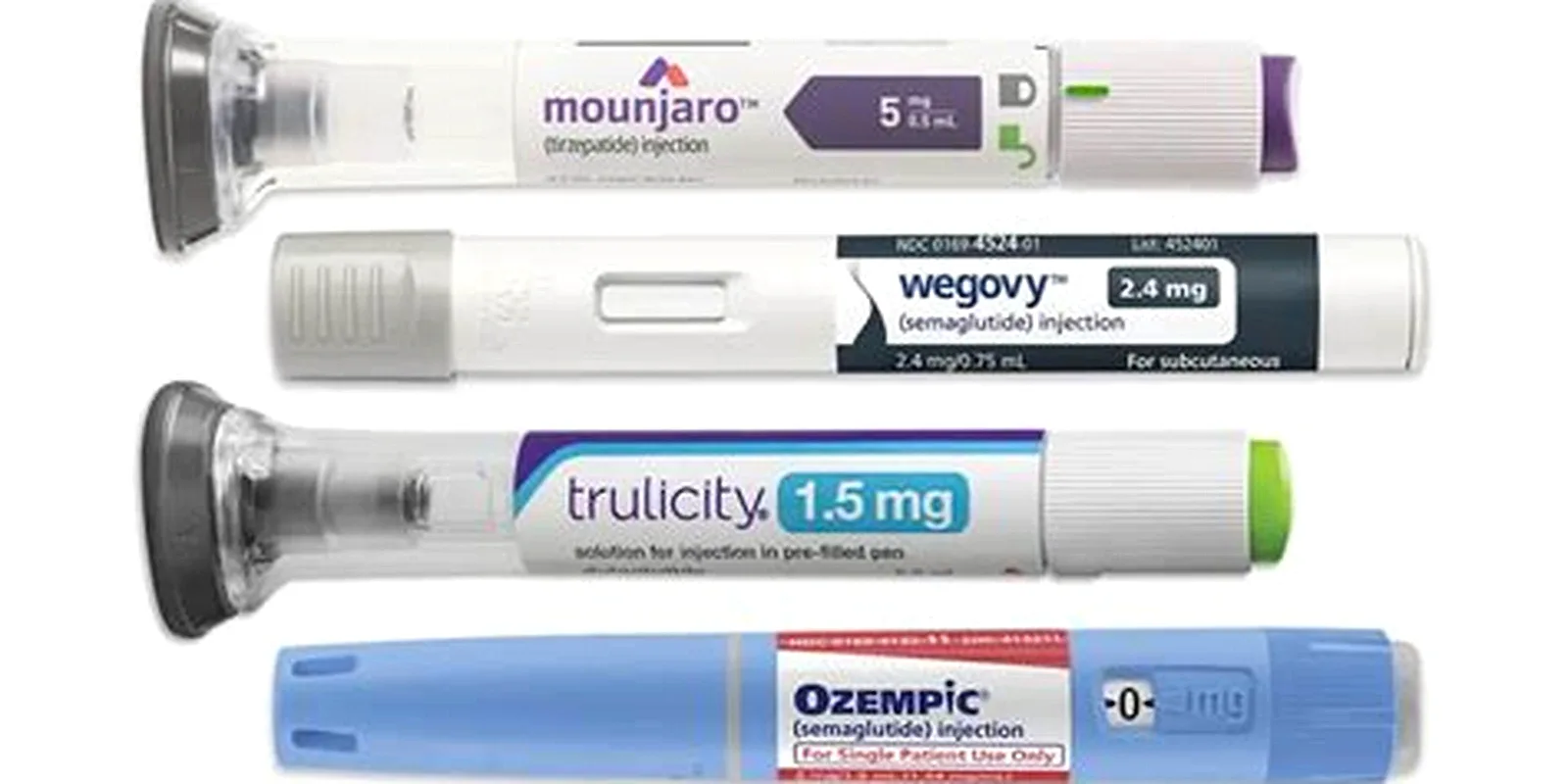
Semaglutide (Ozempic®, Wegovy®, Rybelsus®)
Manufactured by Novo Nordisk. Ozempic was FDA-approved in 2017 for type 2 diabetes; Rybelsus (oral semaglutide) in 2019 for type 2 diabetes; Wegovy in 2021 for chronic weight management in adults with BMI ≥30, or BMI ≥27 with at least one weight-related comorbidity. In 2024 Wegovy received an additional indication for cardiovascular risk reduction in adults with established cardiovascular disease and obesity, based on the SELECT trial.
Clinical efficacy: in the STEP-1 trial (Wilding et al, NEJM 2021), adults on semaglutide 2.4 mg weekly lost a mean 14.9% of body weight at 68 weeks, versus 2.4% on placebo. Both groups received lifestyle counseling.
Tirzepatide (Mounjaro®, Zepbound®)
Manufactured by Eli Lilly. Mounjaro was FDA-approved in May 2022 for type 2 diabetes. Zepbound was approved in November 2023 for chronic weight management, and in December 2024 for obstructive sleep apnea in adults with obesity. Tirzepatide is a dual agonist at the GLP-1 and GIP receptors, making it pharmacologically distinct from pure GLP-1 agonists.
Clinical efficacy: in the SURMOUNT-1 trial (Jastreboff et al, NEJM 2022), adults on tirzepatide 15 mg weekly lost a mean 20.9% of body weight at 72 weeks, the largest weight loss reported for an obesity medication in a published Phase 3 trial.
Liraglutide (Saxenda®, Victoza®)
Manufactured by Novo Nordisk. Victoza was FDA-approved in 2010 for type 2 diabetes; Saxenda in 2014 for chronic weight management. Liraglutide is a once-daily injection. Mean weight loss in the SCALE-Obesity trial was around 5-7% at 56 weeks, smaller than semaglutide or tirzepatide.
Dulaglutide (Trulicity®)
Manufactured by Eli Lilly. FDA-approved in 2014 for type 2 diabetes. Once-weekly injection. No FDA-approved weight management indication.
Exenatide (Byetta®, Bydureon BCise®)
Originally manufactured by Amylin/Eli Lilly, now AstraZeneca. Byetta (twice-daily) was the first-in-class FDA approval in 2005. Bydureon BCise is the extended-release once-weekly formulation, approved in 2012. Less commonly prescribed today, but still in use.
| Brand | Generic | Class | Frequency | FDA indication |
|---|---|---|---|---|
| Ozempic | Semaglutide | GLP-1 receptor agonist | Once weekly (Ozempic, Wegovy); once daily (Rybelsus oral) | Type 2 diabetes (FDA-approved 2017); cardiovascular risk reduction in adults with T2D |
| Wegovy | Semaglutide | GLP-1 receptor agonist | Once weekly (Ozempic, Wegovy); once daily (Rybelsus oral) | Chronic weight management (BMI ≥30, or ≥27 with comorbidity); FDA-approved 2021; CV risk reduction with overweight/obesity and known heart disease |
| Rybelsus | Semaglutide | GLP-1 receptor agonist | Once weekly (Ozempic, Wegovy); once daily (Rybelsus oral) | Type 2 diabetes; oral form, FDA-approved 2019 |
| Mounjaro | Tirzepatide | Dual GIP/GLP-1 receptor agonist | Once weekly | Type 2 diabetes; FDA-approved 2022 |
| Zepbound | Tirzepatide | Dual GIP/GLP-1 receptor agonist | Once weekly | Chronic weight management; FDA-approved November 2023; obstructive sleep apnea in obesity (approved December 2024) |
| Saxenda | Liraglutide | GLP-1 receptor agonist | Once daily | Chronic weight management; FDA-approved 2014 |
| Victoza | Liraglutide | GLP-1 receptor agonist | Once daily | Type 2 diabetes; FDA-approved 2010 |
| Trulicity | Dulaglutide | GLP-1 receptor agonist | Once weekly | Type 2 diabetes; FDA-approved 2014 |
| Byetta | Exenatide | GLP-1 receptor agonist | Twice daily (Byetta) or once weekly (Bydureon) | Type 2 diabetes; FDA-approved 2005 |
| Bydureon BCise | Exenatide | GLP-1 receptor agonist | Twice daily (Byetta) or once weekly (Bydureon) | Type 2 diabetes; extended-release, FDA-approved 2012 |
Find out which GLP-1 option fits your situation
A licensed clinician can review your eligibility for FDA-approved or legally compounded GLP-1 medications via telehealth.
See If You Qualify →Sponsored link. We earn a commission when you sign up. Medical eligibility determined by licensed providers. Disclosure
How GLP-1 Medications Work for Weight Loss
GLP-1 receptor agonists drive weight loss through several converging mechanisms:
- Reduced appetite signaling in the hypothalamic arcuate nucleus and brainstem nucleus tractus solitarius. Patients commonly describe a quieter "food noise" — fewer intrusive thoughts about eating.
- Delayed gastric emptying, which prolongs the feeling of fullness after meals.
- Increased satiety hormone signaling with sustained GLP-1R activation.
- For tirzepatide, additional GIP receptor agonism that may further enhance energy expenditure and lipid metabolism.
The result is a sustained, biologically mediated reduction in caloric intake — typically 20-30% below baseline in trial populations — rather than the willpower-dependent restriction of traditional dieting.
Common Side Effects
The most common GLP-1 side effects are gastrointestinal: nausea, vomiting, diarrhea, constipation, and abdominal pain. In the STEP-1 trial, nausea occurred in 44.2% of participants on semaglutide 2.4 mg, diarrhea in 31.5%, vomiting in 24.8%. Side effects are typically mild-to-moderate, most common during dose escalation, and decrease over time with continued use.
Serious adverse events are uncommon but documented: acute pancreatitis, gallbladder disease, kidney injury (usually from dehydration), severe gastroparesis, hypoglycemia (especially with insulin or sulfonylureas), and rare reports of bowel obstruction. All GLP-1 receptor agonists carry an FDA boxed warning for thyroid C-cell tumors based on rodent studies; they are contraindicated in people with personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2 (MEN-2).
For the full breakdown including emerging issues like NAION (vision), mental health signal reviews, and "Ozempic face," see our complete GLP-1 side effects guide.
Who Should NOT Use GLP-1 Medications
GLP-1 receptor agonists are contraindicated or used with caution in the following situations:
- Personal or family history of medullary thyroid carcinoma (MTC) — contraindicated.
- Multiple endocrine neoplasia syndrome type 2 (MEN-2) — contraindicated.
- Known hypersensitivity to the active ingredient or excipients — contraindicated.
- Pregnancy, planned pregnancy, or breastfeeding — generally avoided; semaglutide should be discontinued at least two months before a planned pregnancy.
- History of pancreatitis — caution, alternative agents may be preferred.
- Severe gastrointestinal disease, gastroparesis, or active bowel obstruction — caution.
- Diabetic retinopathy in patients with type 2 diabetes — risk of acute worsening with rapid HbA1c reduction; close ophthalmologic monitoring advised.
- Type 1 diabetes — GLP-1 RAs are not approved as monotherapy for T1D.
Consult a licensed clinician before starting any GLP-1 medication, especially if any of the above apply.
How to Get a GLP-1 Prescription
There are three common pathways:
- Primary care or endocrinology clinic. Schedule a visit, discuss eligibility (BMI, comorbidities, diabetes status), get labs, and receive a prescription if appropriate. Insurance coverage varies — type 2 diabetes indications are usually covered, weight-management indications often are not.
- Telehealth platform. Complete an online intake, have a video evaluation with a licensed clinician in your state, and receive a prescription sent to a partner pharmacy. Most US telehealth GLP-1 services support either brand-name or legally compounded products.
- Medical weight loss clinic. Often combines lifestyle counseling with prescribing. May be in-person or hybrid.
For step-by-step guidance, see our complete prescription guide.
GLP-1 Cost and Insurance
Without insurance, brand-name GLP-1 medications cost approximately:
- Ozempic: ~$1,000/month
- Wegovy: ~$1,350/month
- Mounjaro: ~$1,100/month
- Zepbound: ~$1,060/month
- Saxenda: ~$1,400/month
- Rybelsus (oral): ~$1,000/month
Manufacturer savings programs (NovoCare, LillyDirect) can reduce out-of-pocket cost to $0-$500 for eligible commercially insured patients. Compounded semaglutide and tirzepatide through telehealth typically cost $150-$500/month, where legally available. Medicare Part D historically excluded weight-loss drugs under a 2003 law, although policy reform is under discussion. For details, see our cost guide and cheapest GLP-1 options articles.
Start with a telehealth eligibility check
A few minutes online tells you whether a licensed clinician can prescribe a GLP-1 medication for your situation — brand-name or legally compounded, depending on what is appropriate.
Start My Online Visit →Sponsored link. We earn a commission when you sign up. Medical eligibility determined by licensed providers. Disclosure